International Fluid Power Inc.
International Fluid Power - Innovative Partners For Global Sourcing Of Fluid Power Products. Your Complete Hydraulic Source in SE Minnesota.
INTERNATIONAL FLUID POWER, INC.
Where Are Peptides Synthesized for Global Supply Chain?
Peptides are crucial in various fields, from pharmaceuticals to biotechnology. Understanding "Where Are Peptides Synthesized" is vital for ensuring a stable supply chain. Dr. Emily Chen, a leading peptide scientist, has stated, "The location of peptide synthesis impacts both quality and accessibility."
Currently, peptide synthesis occurs in multiple regions, including North America, Europe, and Asia. Each region has its unique advantages and challenges. For example, Europe boasts advanced technology but faces stringent regulations. Meanwhile, Asia has lower production costs but may compromise on quality.
With increasing global demand, the industry must address these complexities. Stakeholders must ensure that the synthesis locations align with both quality standards and cost-effectiveness. This brings to light the need for constant evaluation and adaptation. As the landscape shifts, understanding the implications of "Where Are Peptides Synthesized" becomes even more essential for all players in the supply chain.
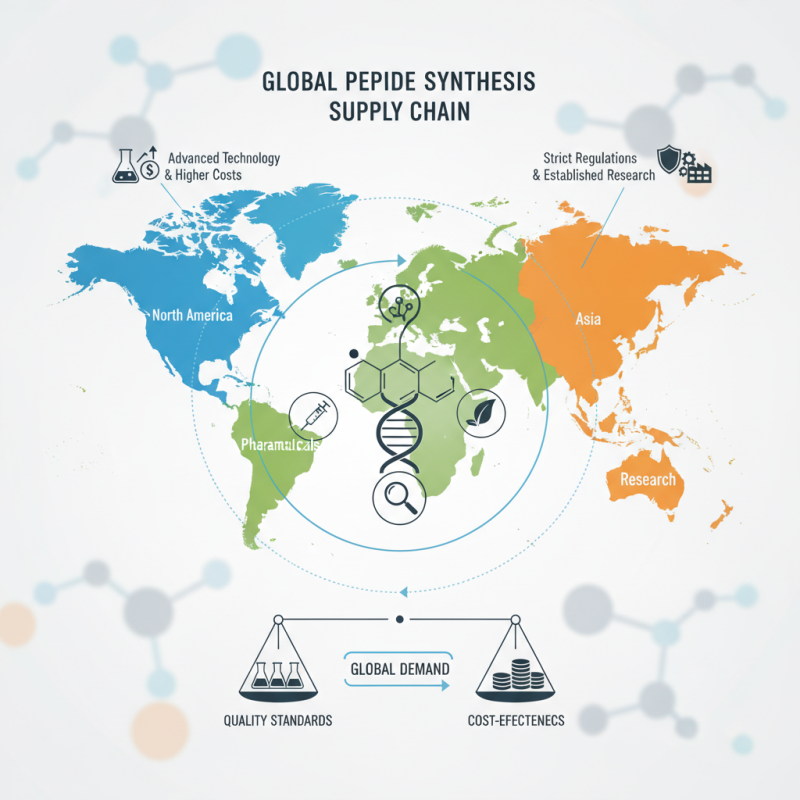
Global Overview of Peptide Synthesis Locations
Peptides are essential in various fields, including medicine and biotechnology. Their synthesis occurs globally in different regions known for advanced research capabilities. Locations like North America, Europe, and parts of Asia are leading players in peptide production. Each region has unique advantages. For instance, North America has a robust infrastructure for scientific research and development.
In Europe, many countries focus on high-quality, specialized peptide synthesis. Cutting-edge technology and skilled professionals enhance their capabilities. Meanwhile, Asia, particularly China and India, offers cost-effective production. These countries benefit from a growing pool of talent and manufacturing expertise. The global supply chain of peptides is complex. It involves a mix of innovation, regional strengths, and resource availability.
Despite advancements, challenges exist within the peptide synthesis landscape. Quality control remains a top concern. The need for consistency can lead to variability in peptide products. Additionally, regulatory hurdles may impact production timelines. It's crucial for stakeholders to address these issues. Continuous improvement and collaboration are essential for maintaining global standards in peptide synthesis.
Key Players in the Global Peptide Supply Chain
The global peptide supply chain plays a critical role in pharmaceuticals and biotechnology. Peptides are synthesized in various facilities worldwide, utilizing advanced technologies. Key players in this sector include manufacturers, contract research organizations, and academic institutions. These entities contribute to research, production, and distribution, creating a complex network.
Manufacturers focus on large-scale production. They often face challenges like cost control and quality assurance. Strict regulatory compliance is essential. Some facilities may struggle with these requirements, leading to inconsistencies in product quality. In contrast, smaller research-focused organizations may offer flexibility in development but often lack the capacity for mass production.
Collaboration is vital in this landscape. Partnerships among key players facilitate innovation and ensure a steady supply of peptides. However, discrepancies in proprietary processes can lead to misunderstandings. As demand continues to rise, adapting to this fast-paced environment remains a challenge. The continuous evolution of technologies shapes the future, yet uncertainty persists about how these advancements will impact the industry.
Global Peptide Supply Chain: Synthesis Locations
Technological Advancements in Peptide Production
Peptide synthesis has seen remarkable technological advancements in recent years. Technologies like solid-phase synthesis and microwave-assisted synthesis are making production faster and more efficient. These innovations allow peptide manufacturers to create complex sequences quickly. The result is a more reliable supply chain that can adapt to the increasing global demand.
Tips: Efficient project planning is vital. Consider investing in advanced synthesis technologies to enhance production capacity. Training your team on the latest techniques may also streamline operations.
Despite the progress, challenges persist. Maintaining quality is critical as production scales up. Often, small errors in synthesis can lead to undesirable outcomes. Regular quality checks are necessary to mitigate these risks.
Tips: Establish a rigorous quality control protocol. Small adjustments in processes can yield significant improvements. Stay informed about best practices in peptide synthesis to ensure success.
Quality Control Measures in Peptide Synthesis
Peptide synthesis is a critical process in the pharmaceutical industry. Ensuring quality control throughout the synthesis process is essential. Studies show that over 30% of synthesized peptides face quality issues, often due to inadequate monitoring. Common challenges include impurities and incorrect sequences. Implementing strict quality control measures can mitigate these issues.
A reliable approach involves the use of high-performance liquid chromatography (HPLC) to detect impurities. Regular testing at each synthesis stage enhances product reliability. According to a recent industry report, companies employing robust quality protocols see a 20% increase in yield. This is vital for maintaining supply chain integrity.
**Tip:** Always validate the source of raw materials. The integrity of peptides relies heavily on starting compounds. Proper documentation is key. Ongoing training for lab personnel on quality practices can further ensure consistency.
Future Trends in Global Peptide Manufacturing
The global demand for peptides is rising rapidly. Industries like pharmaceuticals and biotechnology are expanding. This growth drives the need for effective peptide synthesis methods. Advanced technologies are shaping how peptides are produced, making the process faster and more efficient. Machines and automation streamline production lines. However, not all companies can keep pace with technological changes.
Challenges persist in peptide manufacturing. Quality control remains a significant concern. Ensuring consistency in peptide output is crucial. Companies must invest in R&D to innovate and improve processes. Additionally, sourcing raw materials can pose setbacks. The variability in peptide sequences complicates matters. Stakeholders must collaborate to overcome these hurdles and enhance supply chain reliability.
Looking ahead, sustainability is becoming a focal point. Reducing waste and energy consumption will be essential. The industry must consider environmental impacts. As regulations become stricter, businesses will have to adapt. Future trends will likely emphasize green chemistry and eco-friendly practices. Balancing efficiency with sustainability is no small task. The journey ahead will require resilience and a proactive approach.
Article Source:
You Can Find International Fluid Power Here:
Search For:
Footer #4 Widget
This is an example of a widget area that you can place text to describe a product or service. You can also use other WordPress widgets such as recent posts, recent comments, a tag cloud or more.
© 2025 International Fluid Power, Inc. All Right Reserved.